Unique Conductivity Behavior in Water-in-Salt Electrolytes Driven by Ion Clusters

Understanding and predicting ion transport in aqueous electrolytes are crucial for advanced energy storage and biophysics, and many emergent technologies yet remain elusive.
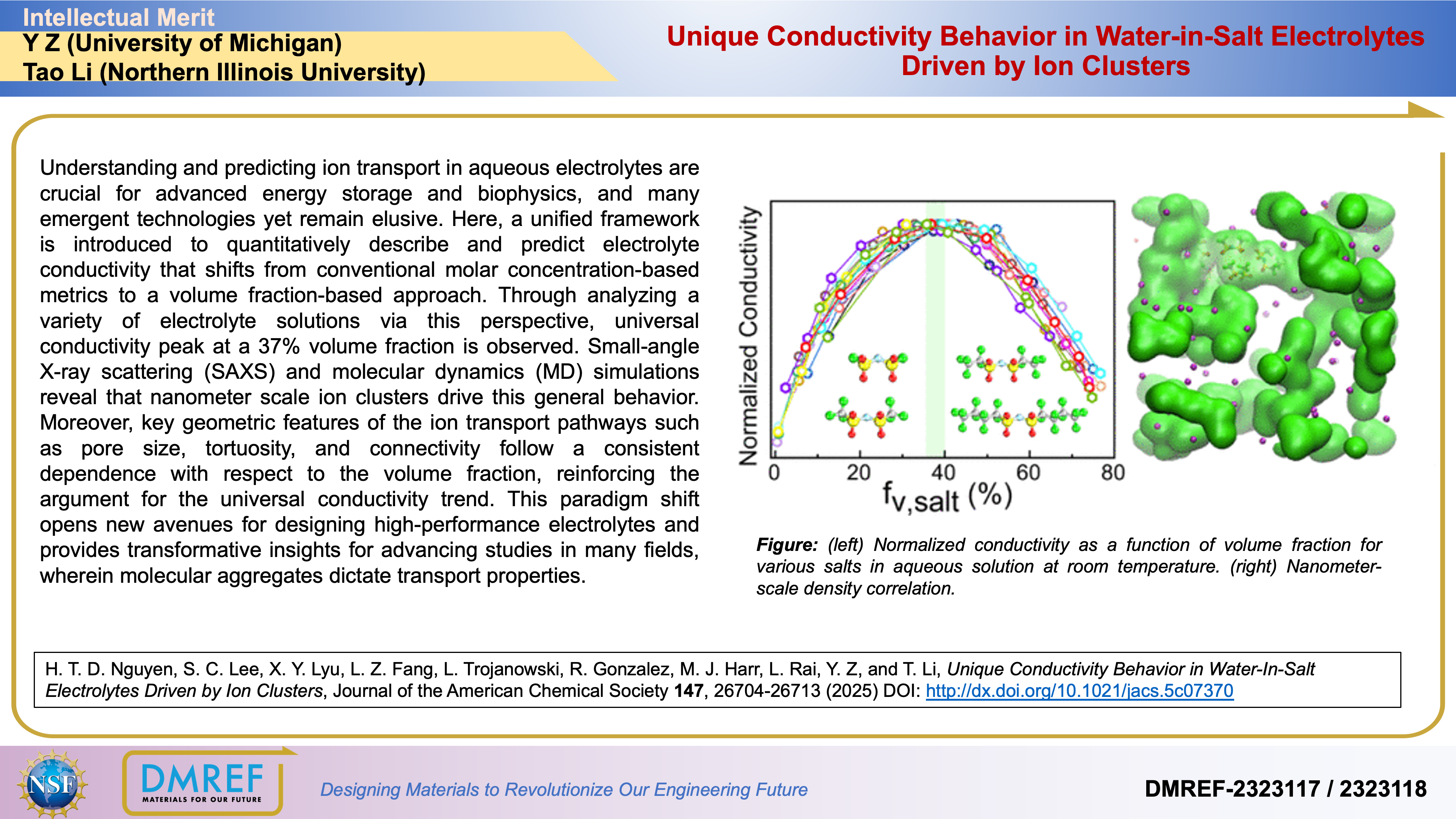
Here, a unified framework is introduced to quantitatively describe and predict electrolyte conductivity that shifts from conventional molar concentration-based metrics to a volume fraction-based approach. Through analyzing a variety of electrolyte solutions via this perspective, universal conductivity peak at a 37% volume fraction is observed. Small-angle X-ray scattering (SAXS) and molecular dynamics (MD) simulations reveal that nanometer scale ion clusters drive this general behavior.
Moreover, key geometric features of the ion transport pathways such as pore size, tortuosity, and connectivity follow a consistent dependence with respect to the volume fraction, reinforcing the argument for the universal conductivity trend. This paradigm shift opens new avenues for designing high-performance electrolytes and provides transformative insights for advancing studies in many fields, wherein molecular aggregates dictate transport properties.